- +1 858 909 0079
- +1 858 909 0057
- [email protected]
- +1 858 909 0079
- [email protected]

Products
Cat. No.
Product Name
Unit Size
Order
Specification
Composition
Magnetic beads charged with Ni2+
Magnetization
~60 EMU/g
Type of Magnetization
Superparamagnetic
Stability
pH 4-11,100% Ethanol, 100% Methanol,8M Urea, 6M guanidine hydrochloride, 20 mM DTT, 20mM EDTA
Concentration
100 mg/ml (1% NiSO4.6H2O)
Binding Capacity
>2mg His-tagged GFP /ml of Beads
Storage
Store at 4ºC upon receipt
Shipping conditions: At ambient temperature
Handling and Storage: Store the kit at 4ºC on arrival.
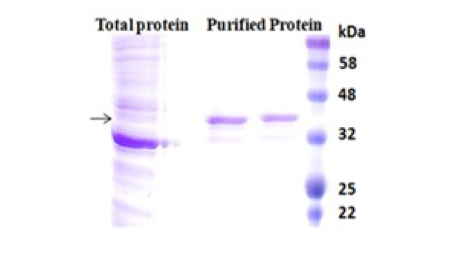
BcMag™ Low Expression His-Tagged Protein Purification Kit uses reducing reagent-compatible magnetic beads to purify his-tagged proteins with nonspecific binding problems such as low expressed protein or the target protein bound with the cellular protein. The beads are manufactured using nanometer-scale superparamagnetic iron oxide as core and entirely encapsulated by a high purity silica shell, ensuring no leaching problems with the iron oxide. Our proprietary chelators are immobilized on their surface and bound firmly with Ni2+ ions, leading to high resistance to reducing reagents. The pure inert silica makes less nonspecific binding due to fewer side chains of the silica materials than other solid matrices such as agarose. Additionally, the beads combine all the advantages of BcMag™ Low expression His-tagged protein purification kit (low costs, simplicity, high specificity, and capacity) and magnetic properties to perform efficient manual or automatic quick high-throughput purification.
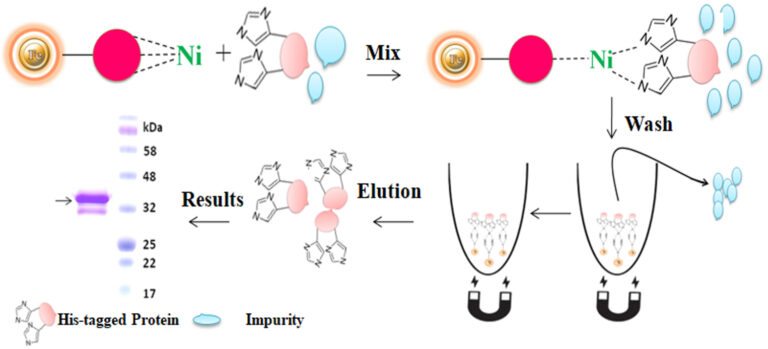
The purification with magnetic microparticles is straightforward. Mix the microparticles with the sample and incubate them with continuous rotation for a sufficient time. During mixing, the beads remain suspended in the sample solution, allowing the target molecules to interact with the immobilized ligand. After incubation, the beads are collected and separated from the sample using a magnet rack. Then the ultrapure His-tagged recombinant proteins are eluted by imidazole.
●
Magnetic beads exhibit less nonspecific binding than porous supports.
●
Stable covalent bond with minimal ligand leakage
●
The beads resist up to 20 mM EDTA and 20 mM reducing reagents without nickel leaching.
●
High protein purity
●
Cost-effective: Eliminates columns, filters, repeat pipetting, and organic reagents.
●
High-throughput: Compatible with many different automated liquid handling systems.
●
Investigating protein structure and function
●
Preparing recombinant proteins for X-ray crystallography
●
Ideal for study of protein interactions with protein or DNA
●
Immunization to raise antibodies against a protein of interest
●
Effective screening protein expression even with crude cell lysates
●
Microscale purification of his-tagged proteins.
A polyhistidin tag, also called 6xHis-tag, His-tagged, and His-tag, is a versatile tool for purifying the highly purified recombinant protein from various expression systems, including bacterial, yeast, plant cell, and mammalian cells systems. The tag comprises six or more consecutive histidine amino acid residues positioned at either N or C terminus of a recombinant protein. Due to its small size, His-tag has several distinctive features, including less immunogenicity, hydrophilic nature, and versatility under native and denaturing conditions. Additionally, it is unnecessary to cleave the tag from the recombinant protein since it rarely perturbs the structure and function of its fusion protein. The purification principle of the His-tag depends on immobilized metal ion affinity chromatography (IMAC).
Nonspecific binding was a potential problem with the purification of his-tagged proteins from various expression systems, especially from the lower expressed recombinant his-tagged proteins or higher expressed proteins interacting with other cellular proteins. Many factors can cause nonspecific binding. Among them, low expressed protein or the tagged protein bound with the endogenous proteins could result in severely nonspecific binding. The binding of His-tagged proteins to the metal ion of the IMAC depends on electric charges. The nonspecific binding occurs when the his-tagged binding sites of resin are only partially bound by the protein of interest due to its low abundance. The rest of the binding sites were non-specifically interacted with other slightly charged proteins such as histidine-rich protein, leading to impurities later on. Multiple lines of evidence prove that reducing reagents and nonionic detergents in the binding and washing buffers could dramatically reduce the highly nonspecific binding and get purer proteins. The most used chelators in IMAC applications are iminodiacetate (IDA) or nitrilotriacetic acid (NTA). These chelators cannot use together with reducing reagents such as DTT (dithiothreitol), β-me ( β-Mercaptoethanol), and TCEP (TRIS (2-carboxyethyl) phosphine) since the reducing reagents can strip off the metal from these resins, resulting in the rapid loss of the protein binding capacity.
Moreover, those chelators are mainly immobilized to the traditional affinity chromatography matrices such as agarose resin or column. These solid matrices make the purification process tedious, time-consuming, unable to handle very tiny samples, and challenging to adapt to the automation system. Bioclone introduces a powerful magnetic bead based IMAC system to address these problems.
1.
Jansen, J-C. (Editor). (2011). Protein Purification: Principles, High-Resolution Methods, and Applications. 3rd edition. Volume 151 of Methods of Biochemical Analysis. John Wiley & Sons, Hoboken, NJ
2.
Bornhorst, J.A. and Falke, J.J. (2000). Purification of Proteins Using Polyhistidine Affinity Tags. Methods Enzymol. 326: 245-254.
3.
Spriestersbach A, Kubicek J, Schäfer F, Block H, Maertens B. Purification of His-Tagged Proteins. Methods Enzymol. 2015;559:1-15. doi: 10.1016/bs.mie.2014.11.003. Epub 2015 May 4. PMID: 26096499.
4.
Zhang C, Fredericks D, Longford D, Campi E, Sawford T, Hearn MT. Changed loading conditions and lysate composition improve the purity of tagged recombinant proteins with tacn-based IMAC adsorbents. Biotechnol J. 2015 Mar;10(3):480-9. doi: 10.1002/biot.201400463. Epub 2014 Oct 31. PMID: 25303209.
5.
Pina AS, et al. (2014) Affinity tags in protein purification and peptide enrichment: An overview. Methods in molecular biology (Clifton, N.J.) 1129: 147-168.
6.
Young CL, et al. (2012) Recombinant protein expression and purification: A comprehensive review of affinity tags and microbial applications. Biotechnol J 7(5): 620-634.
Magnetic Beads Make Things Simple