- +1 858 909 0079
- +1 858 909 0057
- [email protected]
- +1 858 909 0079
- [email protected]

Products
Specification
Composition
Magnetic beads grafted with quaternary amine functional groups.
Magnetization
~45 EMU/g
Type of Magnetization
Superparamagnetic
Effective Density
2.0 g/ml
Stability
Most organic solvents
Strong Anion Exchange beads
1 μm beads: ~3 mg BSA/ ml of Beads
5 μm beads: ~2 mg BSA/ ml of Beads
Storage
Store at 4°C upon receipt.
The utilization of magnetic beads to fractionate proteins or nucleic acids via beads-adsorbent technology as a chromatographic matrix is a highly complex and perplexing process that requires an extensive understanding of the underlying principles. In particular, the widely-used ion exchange chromatography technique involves separating or purifying a target molecule from crude biological materials based on variations in their accessible surface charges. To achieve intact biological activity, this process utilizes very light binding and eluting conditions that demand a significant degree of technical expertise and skill.
At present, strong anion exchange magnetic resins have emerged as a highly effective and cutting-edge alternative to traditional chromatographic procedures that are often time-consuming, complex, and costly. These resins replace materials such as agarose, cellulose, Sepharose, and Sephadex-based columns or resins, which are known for their labor-intensive and time-consuming processing requirements. In column-based procedures, for instance, the lysate is first cleared or centrifuged, after which the supernatant is added to the column, and the membrane or resin is washed with buffer through centrifugation or vacuum manifold. Finally, the required biomolecules are eluted in an adequate volume of buffer. However, this approach may necessitate a significant quantity of hand pipetting, which can discourage differences in target biomolecule yield between experiments and people, leading to significant variability in the results.
Against this backdrop, BcMag™ Strong Anion Exchange (SAX) Magnetic Beads have emerged as a highly effective and innovative solution to these challenges. These uniform magnetic resins have been grafted with a high density of quaternary amine functional group (Strong Anion Exchange) on the surface, which makes them ideally suited for use in high-yield processing of 96 samples in about 20 minutes. By enabling the rapid fractionation of proteins or nucleic acids from complex biological samples (such as serum, plasma, etc.), either manually or automatically, these magnetic beads offer significant advantages over traditional chromatographic techniques. The purified protein can be used in downstream applications such as sample fractionation for 1D and 2D SDS-PAGE, X-ray crystallization, and NMR spectroscopy, making them a highly versatile and powerful tool for modern biological research.
●
Fast and simple – Magnetic beads-based format eliminates columns or filters or a laborious repeat of pipetting or centrifugation.
●
Convenient and expandable – Magnetic format enables high-throughput processing of multiple samples in parallel with many different automated liquid handling systems.
●
Robust – Magnetic beads do not crack or run dry.
●
Low bed volume – Working with small magnetic bead volumes allows for minimal buffer volumes, resulting in concentrated elution fractions.
●
Protein pre-fractionation in cell lysates
●
Optimizing purification conditions for new protein preparation protocols
●
Protein purification and concentration
●
Antibody purification from serum, ascites, or tissue culture supernatant
●
Preparation of samples before 1D or 2D PAGE
●
Phosphopeptide purification before MS analysis
Note: The following protocol is an example of fractionating a protein or peptide sample with BcMag™ SAX magnetic beads. Users are encouraged to choose alternative binding, washing, or elution buffers to get the best results and determine the optimal working conditions based on the protocol and suggestions described in the troubleshooting section. It is critical to match the amount of the beads to the amount of protein in the starting material in all protein purification experiments. It is not only for financial reasons but also because insufficient Strong Anion Exchange resin results in inadequate protein binding in the solution. Too many affinity binding sites will result in the binding of other proteins, making the purification less selective and requiring extra purification steps to achieve pure protein. We recommend performing a titration to optimize the beads used for each application. It is necessary to optimize volumes of elution to avoid unnecessary sample dilution.
Note: Select the appropriate buffer
●
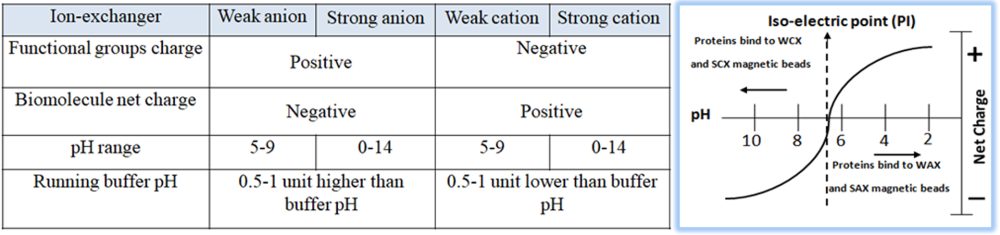
Based on the protein’s pI, empirically calculate the appropriate buffer (pH and salt concentration) for purifying and eluting the protein of interest. In a buffered solution above the protein’s pI, the protein becomes negatively charged (deprotonated) and binds to the positively charged functional groups of an anion exchange resin. To choose the correct buffer for a selected pH, the following is a general rule for selecting a buffer pH:
Anion exchanger — 0.5–1.5 pH units higher than the protein’s pI of interest.
Cation exchanger — 0.5–1.5 pH units lower than the protein’s pI of interest.
A. Equipment
●
Magnetic Rack (for manual operation)
Based on sample volume, the user can choose one of the following Magnetic Racks:
– BcMag™ Magnetic Rack-2 for holding two individual 1.5 ml centrifuge tubes (Cat. No. MS-01);
– BcMag™ Magnetic Rack-6 for holding six individual 1.5 ml centrifuge tubes (Cat. No. MS-02);
– BcMag™ Magnetic Rack-24 for holding twenty-four individual 1.5-2.0 ml centrifuge tubes (Cat. No. MS-03);
– BcMag™ Magnetic Rack-50 for holding one 50 ml centrifuge tube, one 15 ml centrifuge tube, and four individual 1.5 ml centrifuge tubes (Cat. No. MS-04);
– BcMag™ Magnetic Rack-96 for holding a 96 ELISA plate or PCR plate (Cat. No. MS-05).
For larger scale purification, Ceramic Magnets Block for large scale purification (6 in x 4 in x 1 in block ferrite magnet, Applied Magnets, Cat. No. CERAMIC-B8).
●
Corning 430825 cell culture flask for large-scale purification (Cole-Parmer, Cat. No. EW-01936-22)
●
Mini BlotBoy 3D Rocker, fixed speed, small 10″ x 7.5″ platform w/ flat mat (Benchmark Scientific, Inc. Cat. No. B3D1008) or compatible
B. Buffer
●
Binding/Wash Buffer: 25 mM Tris•HCl, pH 8.0. The pH of the Binding/Wash Buffer should be at least one pH unit away from the pI of the target protein or peptide. (Note: For best results, the salt should be ≤ 25mM in the sample).
●
Elution Buffer: 25 mM Tris-HCl pH 8.0. To elute the target protein or peptide from the magnetic beads, the user should optimize elution conditions for individual application by stepwise elution using solutions with increasing salt concentration, i. e., increase stepwise to a final salt concentration of 2.5 M).
General Protocol for using the Strong Anion Exchange Magnetic Beads –
a.
BcMag™ Strong Anion Exchange Magnetic beads preparation
1.
Vigorously shake the bottle until the magnetic resins become homogeneous and transfer an appropriate volume of the magnetic resins from the bottle to a new tube or flask.
Note:
●
Optimize the number of resins used for each application. Insufficient resins will lead to lower yields. Too many beads will cause higher background.
●
Do not allow the resins to sit for more than 3 minutes before dispensing. Resuspend the magnetic beads every 3 minutes.
2.
3.
Repeat step (2) one more time.
4.
Equilibrate the beads by adding ten bead-bed volumes of Binding/Washing buffer and shake it to mix them. Incubate at room temperature with continuous rotation for 2 minutes. Place the tube on the magnetic rack for 1-3 minutes. Remove the supernatant while the tube remains on the rack. The resins are ready for purification.
b.
Purification
1.
Add the equilibrated beads (Step a (4)) to the sample and incubate on Mini BlotBoy 3D Rocker with continuous rotation for 5-10 minutes.
2.
Place the tube on the magnetic rack for 1-3 minutes. Remove the supernatant while the tube remains on the rack. Add ten bead-bed volumes of Binding/Washing buffer and shake it ten times to wash the beads. Again, place the tube on the magnetic rack for 1-3 minutes and remove the supernatant while the tube remains on the rack.
3.
Repeat step (2) six times.
Note:
●
This step is critical to get high pure protein. It may be necessary to wash the beads more than six times for some proteins to reduce the nonspecific binding.
●
Optimize the washing buffer (pH and salt concentration)
Elute protein with an appropriate volume of elution buffer by pipetting up and down 10-15 times or vortex mixer for 5 minutes.
Note:
Determine the optimum elution buffers (pH and salt concentration) and eluting the protein 2-3 times may be necessary.
4.
Elute protein with an appropriate volume of elution buffer by pipetting up and down 10-15 times or vortex mixer for 5 minutes.
Note:
Determine the optimum elution buffers (pH and salt concentration) and eluting the protein 2-3 times may be necessary.
5.
Collect and transfer the supernatant to a new tube.
C. Troubleshooting
Problem
Low yield
Probable Cause
The sample’s ionic strength is high.
Suggestion
Problem
Low yield
Probable Cause
The sample contains interfering detergents.
Suggestion
Problem
The protein failed to elute.
Probable Cause
Ionic interaction is too strong.
Suggestion
Problem
Poor separation
Probable Cause
Carry-over between eluted fractions
Suggestion
Problem
Poor separation
Probable Cause
Proteins or peptides with similar pI to the target protein
Suggestion
Problem
Probable Cause
Suggestions
Low yield
The sample’s ionic strength is high.
The sample contains interfering detergents.
The protein failed to elute.
Ionic interaction is too strong.
Poor separation
Carry-over between eluted fractions
Proteins or peptides with similar pI to the target protein
1.
Wang, F.; Han, G.; Yu, Z.; Jiang, X.; Sun, S.; Chen, R.; Ye, M.; Zou, H. Fractionation of phosphopeptides on strong anion-exchange capillary trap column for large-scale phosphoproteome analysis of microgram samples J. Sep. Sci. 2010, 33 (13) 1879– 87
2.
Alpert AJ, Hudecz O, Mechtler K (2015) Anion-exchange chromatography of phosphopeptides: weak anion exchange versus strong anion exchange and anion-exchange chromatography versus electrostatic repulsion-hydrophilic interaction chromatography. Anal Chem 87: 4704–4711
3.
Han G, Ye M, Zhou H, Jiang X, Feng S, Jiang X, Tian R, Wan D, Zou H, Gu J (2008) Large-scale phosphoproteome analysis of human liver tissue by enrichment and fractionation of phosphopeptides with strong anion exchange chromatography. Proteomics 8: 1346–1361
4.
Liang X, Hu P, Zhang H, Tan W. Hypercrosslinked strong anion-exchange polymers for selective extraction of fluoroquinolones in milk samples. J Pharm Biomed Anal. 2019 Mar 20;166:379-386.
5.
Laha D, Kamleitner M, Johnen P, Schaaf G. Analyses of Inositol Phosphates and Phosphoinositides by Strong Anion Exchange (SAX)-HPLC. Methods Mol Biol. 2021;2295:365-378.
6.
Gu F, Chodavarapu K, McCreary D, Plitt TA, Tamoria E, Ni M, Burnham JJ, Peters M, Lenhoff AM. Silica-based strong anion exchange media for protein purification. J Chromatogr A. 2015 Jan 9;1376:53-63.
7.
Ritorto MS, Trost M, Cook K. Hydrophilic strong anion exchange chromatography for proteomics: what’s the future outlook? Bioanalysis. 2013 Sep;5(18):2219-21.
Get the Latest News and Updates by Email
6393 Nancy Ridge Dr. Suite A
San Diego, CA 92121 USA
Fax: +1-858-909-0057
Get the Latest News and Updates by Email
© 2023 Bioclone Inc. All Rights Reserved.
Magnetic Beads Make Things Simple